AdvanceBio PNGase A (almond), 2 mU
SKU: GKE-5020B-1
Categories: Glycobiology Enzymes, Glycan Analysis
Description
- Enzyme cleaves N-linked glycans from glycopeptides, including glycans with α(1,3)-linked core fucose as seen in insects and plants.
- Incudes reaction buffer:
- 50 mM citrate-phosphate buffer,
- pH 5.0, 0.02% sodium azide.
Specifications
- Enzyme Specific Activity : 5 units/mg
- pH Range : 4-6
- Enzyme Specificity
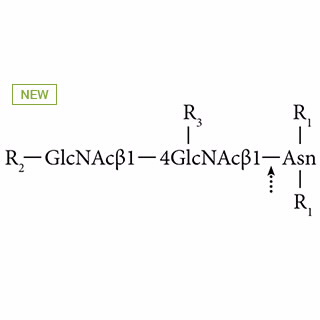
- PNGase A from almonds cleaves N-glycan chains linked to Asparagine from glycopeptides.
- The enzyme hydrolyzes an N(4)-(acetyl-ß D-glucosaminyl) asparagine residue in which the glucosamine residue may be further glycosylated, to yield a (substituted) N-acetyl-ß D-glucosaminylamine and a peptide containing an aspartate residue.
- The enzyme cleaves N-glycans with or without a(1,3)-linked core fucose residues present in insect and plant glycoproteins.
- This differs from PNGase F, which cannot remove N-glycans with a(1,3)-linked core fucose. PNGase A requires glycopeptides as substrates.
- Glycopeptides can be obtained by digesting the glycoprotein with trypsin [1,2] or pepsin.
- Does not act on (GlcNAc)-Asn, because it requires the presence of more than two amino acid residues in the substrate.
- Enzyme Unit Definition : One unit is defined as the amount of enzyme required to hydrolyze 1 µmol ovalbumin glycopeptide per min at pH 5.0 and 37°C
- Enzyme Formulation : Lyophilized powder containing citrate-phosphate buffer, pH 5.0.
- Enzyme Source : Almond. Enzyme also knows as Peptide N-Glycosidase A, Glycopeptidase from almonds, Almond glycoamidase, Peptide-N(4)-(N-acetyl-β glucosaminyl) asparagine amidase.
- Unit : 2 mU
- Molecular Weight : 54.2 and 21.2 kDa
- pH Optimum : 5
*Available in different unit and packing size
GKE-5011A, 2mU https://www.chem.agilent.com/store/productDetail.jsp?catalogId=GKE-5011A&catId=SubCat2ECS_654389
GKE-5011B, 10mU https://www.chem.agilent.com/store/productDetail.jsp?catalogId=GKE-5011B&catId=SubCat2ECS_654389